Abstract
Background
miR-23a, which participates in invasion of pancreatic ductal adenocarcinoma cells into the mesothelial barrier, is a critical regulator in many cancers. It, however, is still unknown whether miR-23a regulates pancreatic cell proliferation and apoptosis or not.
Aims
We sought to investigate the role of miR-23a in regulation of pancreatic cell proliferation and apoptosis.
Methods
miRNA, mRNA, and protein expressions were determined by qRT-PCR and Western blot, respectively. Dual-luciferase reporter assay was used in detection for binding ability of miR-23a to APAF1. Ectopic miR-23a and APAF 1 were introduced to pancreatic cells, and their roles in proliferation and apoptosis were detected by MTT, colony formation, and apoptosis assays, respectively.
Results
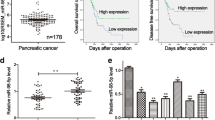
Up-regulation of miR-23a and down-regulation of APAF 1 were found in pancreatic ductal cancer, respectively. miR-23a significantly inhibited the luciferase activity by targeting APAF 1 3′UTR. Ectopic miR-23a significantly suppressed the APAF 1 gene expression in pancreatic cancer cells. Similar to siAPAF1, miR-23a significantly promoted pancreatic cancer cell proliferation and repressed apoptosis. Furthermore, miR-23a inhibitor and exogenous APAF 1 could recover the effects.
Conclusions
It is suggested that miR-23a, acting as an oncogenic regulator by directly targeting APAF 1 in pancreatic cancer, is a useful potential biomarker in diagnosis and treatment of pancreatic cancer.




Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30.
Chen W, Zheng R, Zhang S, et al. Report of incidence and mortality in China cancer registries, 2009. Chin J Cancer Res. 2013;1:10–21.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Kumar-Sinha C, Wei I, Simeone DM. Emerging frontiers in pancreatic cancer research: elaboration of key genes, cells and the extracellular milieu. Curr Opin Gastroenterol. 2012;8:516–522.
Hamacher R, Schmid RM, Saur D, Schneider G. Apoptotic pathways in pancreatic ductal adenocarcinoma. Mol Cancer. 2008;7:64.
Pass HI, Beer DG, Joseph S, Massion P. Biomarkers and molecular testing for early detection, diagnosis, and therapeutic prediction of lung cancer. Thorac Surg Clin. 2013;23:211–224.
Yong FL, Wang CW, Roslani AC, Law CW. The involvement of miR-23a/APAF1 regulation axis in colorectal cancer. Int J Mol Sci. 2014;15:11713–11729.
Wang N, Zhu M, Tsao SW, Man K, Zhang Z, Feng Y. MiR-23a-mediated inhibition of topoisomerase 1 expression potentiates cell response to etoposide in human hepatocellular carcinoma. Mol Cancer. 2013;12:119.
Piepoli A, Tavano F, Copetti M, et al. Mirna expression profiles identify drivers in colorectal and pancreatic cancers. PLoS One. 2012;7:e33663.
Listing H, Mardin WA, Wohlfromm S, Mees ST, Haier J. MiR-23a/-24-induced gene silencing results in mesothelial cell integration of pancreatic cancer. Br J Cancer. 2014;. doi:10.1038/bjc.2014.587.
Wu CC, Bratton SB. Regulation of the intrinsic apoptosis pathway by reactive oxygen species. Antioxid Redox Signal. 2013;19:546–558.
Umetani N, Hoon DS. Frequent LOH at chromosome 12q22-23 and Apaf-1 inactivation in glioblastoma. Brain Pathol. 2003;13:431–439.
Fu WN, Bertoni F, Kelsey SM, et al. Role of DNA methylation in the suppression of Apaf-1 protein in human leukaemia. Oncogene. 2003;22:451–455.
Chen Q, Xu J, Li L, et al. MicroRNA-23a/b and microRNA-27a/b suppress Apaf-1 protein and alleviate hypoxia-induced neuronal apoptosis. Cell Death Dis. 2014;5:e1132.
Lian S, Shi R, Bai T, et al. Anti-miRNA-23a oligonucleotide suppresses glioma cells growth by targeting apoptotic protease activating factor-1. Curr Pharm Des. 2013;19:6382–6389.
Li B, Sun M, Gao F, et al. Up-regulated expression of miR-23a/b targeted the pro-apoptotic Fas in radiation-induced thymic lymphoma. Cell Physiol Biochem. 2013;32:1729–1740.
Mi S, Lu J, Sun M, et al. MicroRNA expression signatures accurately discriminate acute lymphoblastic leukemia from acute myeloid leukemia. Proc Natl Acad Sci USA. 2007;104:19971–19976.
Gottardo F, Liu CG, Ferracin M, et al. Micro-RNA profiling in kidney and bladder cancers. Urol Oncol. 2007;25:387–392.
Saumet A, Vetter G, Bouttier M, et al. Transcriptional repression of microRNA genes by PML-RARA increases expression of key cancer proteins in acute promyelocytic leukemia. Blood. 2009;113:412–421.
Kozaki K, Imoto I, Mogi S, Omura K, Inazawa J. Exploration of tumor-suppressive microRNAs silenced by DNA hypermethylation in oral cancer. Cancer Res. 2008;68:2094–2105.
Corvaro M, Fuoco C, Wagner M, Cecconi F. Analysis of apoptosome dysregulation in pancreatic cancer and of its role in chemoresistance. Cancer Biol Ther. 2007;6:209–217.
Hoskins JW, Jia J, Flandez M, et al. Transcriptome analysis of pancreatic cancer reveals a tumor suppressor function for HNF1A. Carcinogenesis. 2014;25:2670–2678.
Chhabra R, Dubey R, Saini N. Cooperative and individualistic functions of the microRNAs in the miR-23a ~27a ~24-2 cluster and its implication in human diseases. Mol Cancer.. 2010;9:232.
Liu X, Ru J, Zhang J, et al. miR-23a targets interferon regulatory factor 1 and modulates cellular proliferation and paclitaxel-induced apoptosis in gastric adenocarcinoma cells. PLoS One. 2013;8:e64707.
Li X, Liu X, Xu W, et al. c-MYC-regulated miR-23a/24-2/27a cluster promotes mammary carcinoma cell invasion and hepatic metastasis by targeting Sprouty2. J Biol Chem. 2013;288:18121–18133.
Permuth-Wey J, Chen YA, Fisher K, et al. A genome-wide investigation of microRNA expression identifies biologically-meaningful microRNAs that distinguish between high-risk and low-risk intraductal papillary mucinous neoplasms of the pancreas. PLoS One. 2015;10:e0116869.
Acknowledgments
This work was supported by the National Natural Science Foundations of China (81172577, 81372876 and 81301767).
Conflict of interest
Fu WN is currently receiving two Grants from National Natural Science Foundations of China (81172577 and 81372876). Liu N is currently getting a Grant from National Natural Science Foundations of China (81301767). For the remaining authors, none declared the conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, N., Sun, YY., Zhang, XW. et al. Oncogenic miR-23a in Pancreatic Ductal Adenocarcinogenesis Via Inhibiting APAF1 . Dig Dis Sci 60, 2000–2008 (2015). https://doi.org/10.1007/s10620-015-3588-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3588-x